Ernest Rutherford 1871 - 1937
Rutherford was born in 1871 in Nelson, New Zealand. He had a good understanding of maths and did original work on radio waves at Canterbury College, Christchurch. He won a scholarship to go to the Cavendish Laboratory in Cambridge, where J. J. Thompson, who had discovered the electron, supervised him. Rutherford studied the conduction of electricity through gases and discovered alpha and beta particles. Soon he was offered a professorship at McGill University, Montreal and had enough money to marry his childhood sweetheart, Mary Newton. In Montreal, with Frederick Soddy, he showed that radioactivity was a process in which atoms of one element decayed spontaneously into atoms of another.
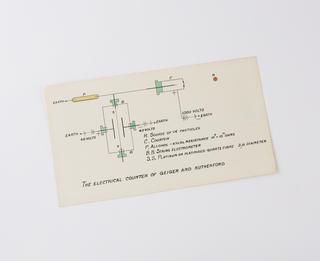
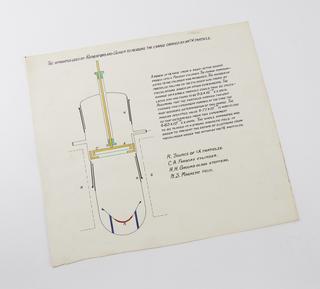
The scientific community was impressed and Rutherford was elected a Fellow of the Royal Society. In 1907, he was invited to take over the chair of physics at the University of Manchester, at the express wish of the outgoing Professor, Arthur Schuster. In 1909, Hans Geiger and Ernest Marsden, researchers working with Rutherford at Manchester, bombarded gold foil with alpha particles from helium nuclei. Most of the particles went straight through as expected but a few bounced back. Rutherford was astounded, remarking: 'It was as though you had fired a fifteen-inch shell at a piece of tissue paper and it had bounced back and hit you.'
In 1911, to explain this, Rutherford proposed a new structure for the atom. He imagined the atom as a miniature solar system with a nucleus at its centre and electrons orbiting around. Although the nucleus was only a million-millionth the volume of the atom, it had over 99.9% of the mass. The nucleus also had a positive electric charge to balance the negative electric charge of the electrons around it. Rutherford was knighted in the 1914 New Year’s Honours List.
Work on radiation ceased during the First World War and Rutherford led the Allied research into the detection of submarines. However, in 1917 Rutherford and his technician, William Kay, set up an experiment using alpha particles as projectiles with which to hit nitrogen atoms. In a long series of exhausting tests, they showed that the result was the liberation of the nucleus of a hydrogen atom. Rutherford had achieved the transmutation of one element into another. Surprisingly, there was very little interest in either the scientific or popular press when he published his results in 1919.
Soon after, he moved to head the Cavendish Laboratory in Cambridge. There, he worked out ways of accelerating particles so that they could be used to explore the secret recesses of matter. He became President of the Royal Society in 1925 and Lord Rutherford in 1931. His researchers carried out further important work: in 1932, James Chadwick found the neutron, and John Cockroft and Ernest Walton transformed lithium atoms into helium by bombarding them with protons. By then, nuclear research was becoming an activity that required teams of researchers and highly specialised, expensive equipment.
Rutherford died suddenly on 19 October 1937 and was buried at Westminster Abbey.